Press Releases

JAMSTEC
Air pollution causes high iron solubility in anthropogenic aerosols during atmospheric transport: Iron fertilization of the open ocean
1. Key points
- ◆
- An international research project on atmospheric iron has been undertaken in the framework of the United Nations Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection (GESAMP).
- ◆
- It was confirmed that anthropogenic iron in aerosols from air pollution is dissolved more quickly in water than natural iron during atmospheric transport.
- ◆
- The results of this study suggest that anthropogenic iron oxides play an important role in providing nutrients to marine ecosystems in the open ocean. It is expected that future impact assessments will contribute to marine environmental conservation policies.
2. Overview
Akinori Ito of the Project Team for Advanced Climate Modeling at the Japan Agency for Marine-Earth Science and Technology (JAMSTEC) and his colleagues collected model results and data from the international community (GESAMP, *1) to statistically analyze prediction results from multiple global atmospheric chemical transport models (*2) and observation data from various regions of the world’s ocean. The results of the team’s analyses showed that anthropogenic iron (*3) changes its chemical properties as it is transported through the atmosphere to the sea. Moreover, this is a primary factor in a large amount of water-soluble iron being incorporated into microparticles (aerosols) over the open ocean.
Iron is a necessary nutrient for marine phytoplankton; in areas of the ocean where iron is scarce, dissolved iron, supplied via airborne aerosols (*4), becomes especially important. Previously, only those naturally occurring iron oxides that have demonstrated low solubilities (*5) were considered as atmospheric sources of iron to the ocean. However, in recent years, observations have indicated that aerosols impacted by airborne pollutants have high iron solubility. Nonetheless, the observational data alone do not provide clarity with respect to why aerosols demonstrate such high iron solubility in the open ocean. Additionally, iron solubility prediction results from mathematical models have shown a large degree of scatter, so conclusions regarding the causes of high aerosol iron solubility differ according to the methods employed.
In this study, JAMSTEC and international scientists combined results from multiple mathematical models with observation data to statistically clarify the cause of the high iron solubility in the marine atmosphere. As a result, it was found that the degree of and changes in solubility were most faithfully reproduced in models that predicted an increase in anthropogenic iron solubility due to the high acidity of atmospheric aerosols during atmospheric transport (Fig. 1). The results further demonstrated that in combination with aerosols from air pollutants, anthropogenic iron becomes highly water-soluble in very acidic solutions due to photochemical reactions. (Fig. 2). The study showed that unlike the conventionally considered, naturally occurring iron oxides, anthropogenic iron oxides play a crucial role in providing nutrients to marine organisms that live far from land, and thus from sedimentary sources of oceanic iron. Future assessments of the effects that the burning of fossil fuels have on marine food chains (and therefore ecosystems) founded upon phytoplankton (primary producers) are expected to contribute to the creation of more effective environmental conservation plans. This study reveals the considerable potential to incorporate extensive biogeochemical–climatic interactions between the atmosphere and ocean ecosystems in Earth system models (ESMs) to aid in marine management and climate policy formation.
This study was based on research supported by the Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research (grant no. JP16K00530) and by the Integrated Research Program for Advancing Climate Models of the Ministry of Education, Culture, Sports, Science and Technology and its Regional Theme B: “Explaining the Carbon ssCycle, Climate Sensitivity, Tipping Elements, and More.”
Further, this study resulted from the deliberations of the United Nations Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection (GESAMP) Working Group 38, “The Atmospheric Input of Chemicals to the Ocean.”
The results were published by Science Advances online on May 2, 2019 (JST).
https://advances.sciencemag.org/content/5/5/eaau7671/
- 1 Project Team for Advanced Climate Modeling, Yokohama Institute for Earth Sciences, Japan Agency for Marine–Earth Science and Technology, Japan
- 2 Institute for Marine and Atmospheric Research, Utrecht University, The Netherlands
- 3 Institute for Environmental Research and Sustainable Development, National Observatory of Athens, Greece
- 4 Environmental Chemical Processes Laboratory, Department of Chemistry, University of Crete, Greece
- 5 Department of Earth and Atmospheric Sciences, Cornell University, USA
- 6 Centre for Ocean and Atmospheric Sciences, School of Environmental Sciences, University of East Anglia, UK.
- 7 Geosciences Division, Physical Research Laboratory, Ahmedabad, India
- 8 Department of Earth & Environmental Science, Rutgers University, USA
- 9 Department of Earth, Ocean, and Atmospheric Science, Florida State University, USA
- 10 Skidaway Institute of Oceanography, University of Georgia, USA
- 11 Institute for Marine and Antarctic Studies, University of Tasmania, Australia
- 12 Laboratoire d’Océanographie de Villefranche, Centre National de la Recherche Scientifique, Sorbonne Université, France
- 13 Department of Marine, Earth, and Atmospheric Sciences, North Carolina State University, USA
- 14 Ames Research Center, National Aeronautics and Space Administration, USA
- 15 Climate & Atmospheric Science Department, Environmental Science Division, Argonne National Laboratory, USA
- 16 Department of Atmospheric and Oceanic Sciences, University of California Los Angeles, USA
- 17 Laboratory of Atmospheric Processes and Their Impacts, School of Architecture, Civil and Environmental Engineering, École Polytechnique Fédérale de Lausanne, Switzerland
- 18 Institute of Chemical Engineering Sciences, Foundation for Research and Technology – Hellas, Greece
- 19 Departments of Oceanography and Atmospheric Sciences, Texas A&M University, USA
- *1
- GESAMP:
An international group of independent scientific experts that provides advice to the United Nations on science of marine environmental protection, officially named the “Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection.” The goal of this group is to conduct scientific assessments of the impacts of human activity on the ocean from a standpoint independent of those of nations or international organizations, as well as to provide advice for policy creation.
- *2
- Atmospheric chemical transport model:
A mathematical model that accounts for the photochemical transformation of aerosols during atmospheric transport, and uses supercomputers to calculate the distributions of various airborne substances, as well as how such distributions may change over time. These models are used not only to explain the causes behind past changes in particle distributions, but also to assess the impacts of emissions standards on various chemical substances on the future atmospheric environment and climate.
- *3
- Anthropogenic iron:
The portion of iron present in atmospheric microparticles that are emitted by human activity, such as the combustion of fossil fuels.
- *4
- Dissolved iron:
Iron dissolved in water. As the atmosphere and the surface ocean are oxidizing environments, dissolved iron is considered to exist in a stable state in aerosols and seawater, primarily as organic complexes.
- *5
- Iron solubility:
Iron solubility expresses the proportion of iron that dissolves in solutions (e.g., water) relative to the iron present in microparticles. As almost no organisms are able to use the iron that is resistant to dissolution in seawater, this iron instead sinks to the ocean floor; therefore, it is an important index when assessing the impacts of aerosols on marine ecosystems.
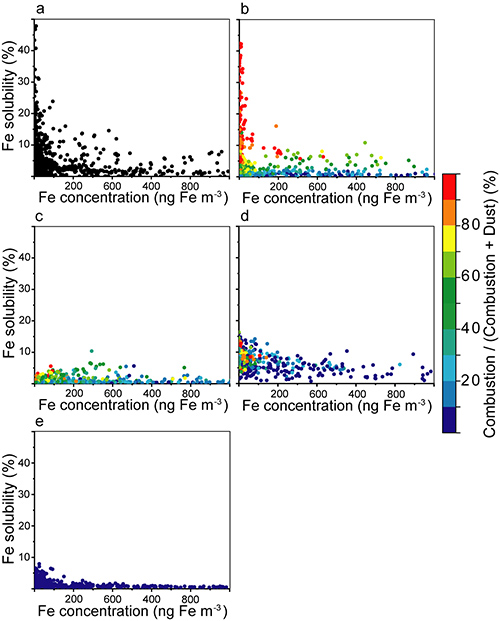
Fig. 1. Relationship between aerosol iron concentration (ng m–3) and iron solubility (%) for the (a) field data and (b) IMPACT, (c) TM4-ECPL, (d) CAM4, and (e) GEOS-Chem models. The color of the prediction results for each mathematical model represents the proportion of dissolved anthropogenic iron vs. all dissolved iron present in the aerosols, with warm colors indicating higher proportions.
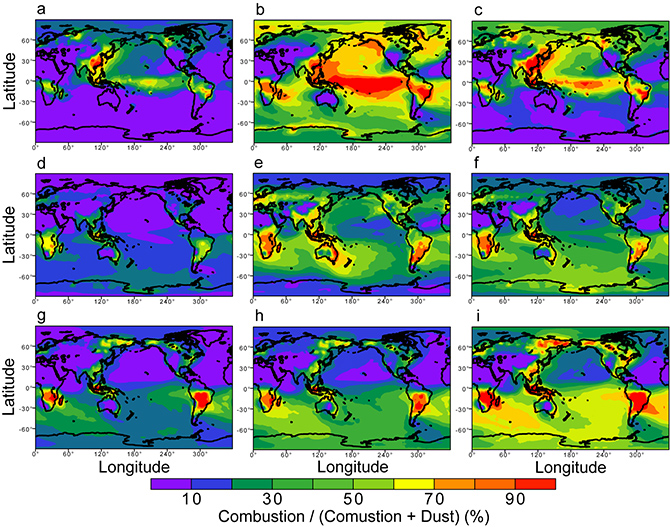
Fig. 2. Proportion of (a, d, and g) anthropogenic iron in all aerosol iron particles, (b, e, and h) dissolved anthropogenic iron, accounting for photochemical reactions in the atmosphere, and (c, f, and i) anthropogenic dissolved iron particles, not accounting for photochemical reactions in the atmosphere. Results are those yielded by the (a, b, and c) IMPACT, (d, e, and f) TM4-ECPL, and (g, h, and i) CAM4 models, with warmer colors indicating higher proportions.
Contacts
- (For this study)
- Akinori Ito, Senior Scientist, Earth System Model Development and Application Group, Research Center for Environmental Modeling and Application, Research Institute for Global Change
- (For press release)
- Public Relations Section, Marine Science and Technology Strategy Department