Press Releases
JAMSTEC
Hokkaido University
The University of Tokyo
Insight into anaerobic methanotrophy from 13C/12C- amino acids
and 14C/12C-ANME cells in seafloor microbial ecology
Overview
A research group led by Dr. Yoshinori Takano from the Department of Biogeochemistry, the Japan Agency for Marine-Earth Science and Technology (JAMSTEC; President: Asahiko Taira) investigated highly efficient methane-consuming microbes (*1) living on the deep seafloor of the Black Sea. His group discovered that biochemical reactions related to amino acid metabolism result in the elongation of 12C-enriched carbon during amino acid biosynthesis. This research project was carried out in collaboration with Hokkaido University and the University of Tokyo in Japan, and Federal Institute for Geosciences and Natural Resources in Germany.
Although it is well known that a large amount of methane is produced in the global sub-seafloor environment, only a portion of it is released into the atmosphere. Twenty years ago, pioneering scientists found 12C-enrichment in archea-derived organic matter and suggested that anaerobic methanotrophic archaea (ANME) beneath the seabed actively consume methane by anaerobic methane oxidation. However, molecular dynamics underlying biochemical reactions within ANME cells and the mechanism of the 12C-enrichment process have remained enigmatic over the last 20 years.
To investigate the metabolic pathway of ANME, Dr. Takano’s research group performed precise chemical analyses using molecular-specific stable isotope mass spectrometry for determining 13C/12C and cellular-specific accelerator mass spectrometry for determining 14C/12C. Compound-specific analysis of ANME-dominated microbial mats showed a significant 13C-depletion trend in association with increasing carbon numbers in protein-derived amino acid families (e.g., the pyruvate family in the order of alanine, valine, isoleucine and leucine was down to −114‰). The results clearly demonstrate that when ANMEs consume methane, 12C is preferentially incorporated into the formation of the “lightest”12C-amino acids (i.e., 13C-depleted amino acids).
The greenhouse effect by methane (*2) is 25 times higher than that by carbon dioxide. Therefore, the role of ANME is very important for global warming. Dr. Takano said that the methane consumption process of ANMEs revealed by this study plays a significant role in stabilizing the global climate.
This work was supported by JSPS KAKENHI Grant Numbers 16H04083 and JP15H05322.
The above results were published in Scientific Reports on September 24, 2018 (JST). Please see, https://www.nature.com/articles/s41598-018-31004-5.
Title: Insight into anaerobic methanotrophy from 13C/12C- amino acids and 14C/12C-ANME cells in seafloor microbial ecology
Authors: Yoshinori Takano1,2, Yoshito Chikaraishi1,3, Hiroyuki Imachi2,4, Yosuke Miyairi5, Nanako O. Ogawa1,2, Masanori Kaneko1,6, Yusuke Yokoyama1,5, Martin Krüger7, Naohiko Ohkouchi1,2
Affiliations: 1. Department of Biogeochemistry, JAMSTEC 2. Research and Development Center for Marine Resources, JAMSTEC 3. Institute of Low Temperature Science, Faculty of Environmental Earth Science, Hokkaido University 4. Department of Subsurface Geobiological Analysis and Research, JAMSTEC 5. Atmosphere and Ocean Research Institute, The University of Tokyo 6. National Institute of Advanced Industrial Science and Technology 7. Federal Institute for Geosciences and Natural Resources (Germany)
*1 ANaerobic Methanotrophic (ANME) archaea: They cause methane (CH4) to undergo a microbiological oxidation reaction with an oxidizing agent such as oxygen, nitric acid, or sulfuric acid. In the presence of oxygen, the process is referred to as aerobic methane oxidation, whereas in the absence of oxygen, it is referred to as anaerobic methane oxidation. This study pertains to the latter. For example, when methane is combined with sulfuric acid, the following anaerobic methane oxidation reaction occurs:
CH4 + SO42- → HCO3- + HS- + H2O
ANME archaea play a central role in such reactions. Ecology is the study of the interaction between (micro) organisms and their environment.
*2 Greenhouse gas: If the global warming coefficient of carbon dioxide is 1, then that of methane is 25. In general, methane at the ocean bottom is used up through the anaerobic methane oxidation reaction before it can dissipate upwards to the seafloor surface. As a result, methane is not observed to gush out at the Black Sea floor. The presenters have previously revealed direct evidence for microbiological methane production occurring deep under the seafloor.
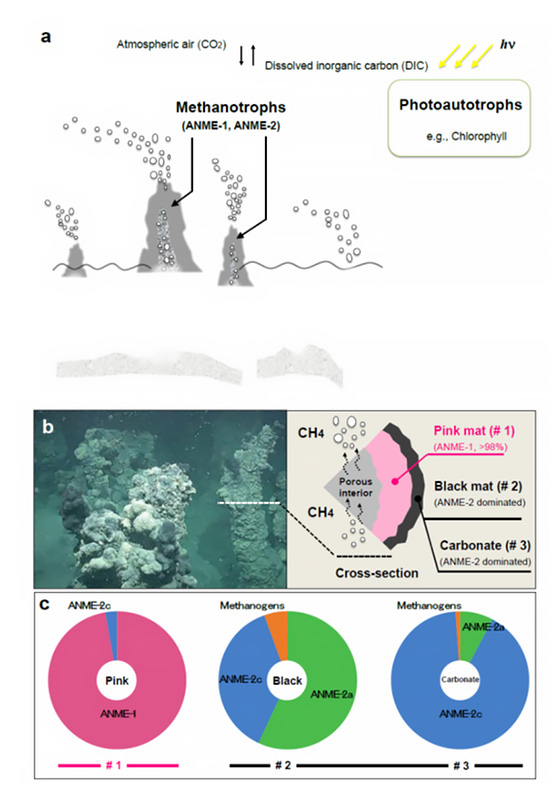
Figure 1. ANME-dominated microbial mats from the Black Sea. (a) Benthic methane seep environment of the Black Sea. (b) Interior and exterior sections of chimney structure habitat location (image capture during the expedition) showing a pink mat (ANME-1-dominated), black mat, and carbonate precipitate (ANME-2-dominated). Photo credit: R/V Meteor cruise M72/1 science party (taken by M. Krüger). (c) The community structures of the ANME samples were determined by methyl coenzyme M reductase A (mcrA) gene-based clone analyses of the pink mat, black mat, and carbonate samples. ANME-1 and ANME-2 dominate the pink and black sections, respectively.
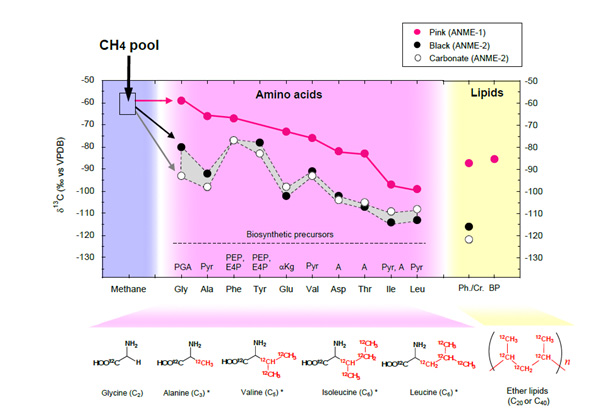
Figure 2. δ13C values of amino acids and lipids extracted from ANME mats in the Black Sea. Chemical structure and 13C-depletion of neutral amino acids glycine, alanine, valine, isoleucine, and leucine with carbon numbers (Cn) up to C6. The asterisks (*) represent pyruvate amino acid family members. Abbreviations: PGA, phosphoglyceric acid; Pyr, pyruvate; A, aspartic acid; αkg, α-ketoglutarate; PEP + E4P, phosphoenolpyruvate + erythrose-4-phosphate. The carbon isotopic composition of the ANME-2-dominated black mat and carbonate included the major archaeal C20 isoprenoid (<−116‰, vs. VPDB)
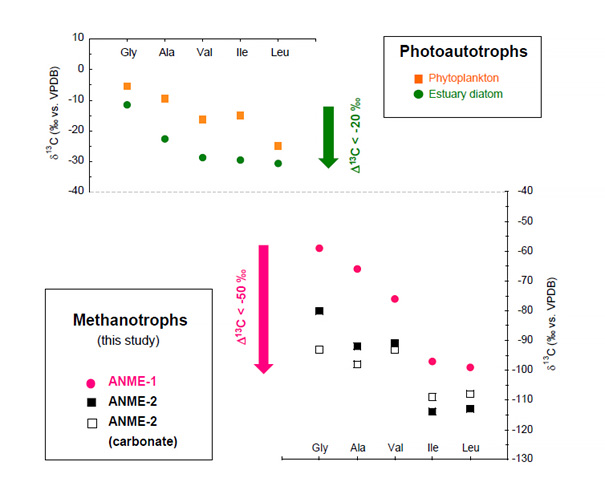
Figure 3. Carbon isotope ratios (13C/12C) of amino acids. Carbon isotopic composition of neutral amino acids in photoautotrophs (upper diagram) and methanotrophs (lower diagram). 13C-depletion proceeds through carbon elongation for C2-glycine, C3-alanine, C5-valine, C6-isoleucine, and C6-leucine. Δ13C is defined as the difference of δ13C during these target molecules.

Figure 4. Examples of ANME-1 and ANME-2 sampled from the deep seafloor.
The scale bar is 3 cm, and the color scale indicates R for red, G for green, and B for blue.
Contacts:
- (For this study)
- Yoshinori Takano, Senior Scientist, Department of Biogeochemistry、JAMSTEC
- Yoshito Chikaraishi, Professor, Institute of Low Temperature Science, Faculty of Environmental Earth Science, Hokkaido University
- Yusuke Yokoyama, Professor, Atmosphere and Ocean Research Institute, The University of Tokyo
- (For press release)
- Tsuyoshi Noguchi, Manager, Press Division, Public Relations, JAMSTEC
- Naoki Namba, Media Officer, Institute for International Collaboration,
Hokkaido University - Yoko Ogawa, Public Relations Office, Atmosphere and Ocean Research Institute,
The University of Tokyo