
July 29, 2008
Japan Agency for Marine-Earth Science and Technology
Extending a Record of Upper Temperature Limit for Life
by a newly-developed high-pressure cultivation technique
Outline
Ken Takai, Program Director of Subground Animalcule Retrieval (SUGAR) Program, Extremobiosphere Research Center, Japan Agency for Marine-Earth Science and Technology (JAMSTEC: Yasuhiro Kato, President) discovered that a hyperthermophilic methanogen isolated from a deep-sea hydrothermal field in the Indian Ocean can grow at up to a high temperature of 122°C under the in situ high pressures, with use of a newly-developed cultivation technique.
The upper temperature limit for life have been reported to be 113°C as a reproducible record (reported record is 121°C). Now the limit of life activity proved to be 122°C. This finding contributes to understanding for not only extent of biosphere on Earth but also limit of surviving conditions of extraterrestrial life forms in space environment.
Moreover, with cooperation of University of the Ryukyus and Hokkaido University, it is discovered that methane produced by the hyperthermophilic methanogen was isotopically abnormal, that means highly 13C-enriched biological methane. This achievement revolutionizes the concept written in geochemistry textbooks that isotopically light methane is derived from the microbiological activities while isotopically heavy methane is abiotic origins.
These achievements was appeared in online version of the Proceedings of the National Academy of Sciences of the USA (PNAS) on July 29.
Background
The history of extending the upper temperature limit (UTL) for life started in 1960s. It is also the history of understanding of habitability on Earth and other planets and moons (Fig.1).
Reproducible record of upper temperature limit for life was believed to be 113°C that had been reported in 1997 as for hyperthermophilic archaeon Pyrolobus fumarii (Blochl et al., 1997). In 2003, the isolation of a hyperthermophilic archaeon strain 121 capable of growth at up to 121°C had been reported (Kashefi and Lovely 2003), but unfortunately it has been never verified yet by other researchers..
These hyperthermophilic archaea are chemolithoautotrophic microorganisms (*1) derived from deep-sea hydrothermal systems. They need to be cultured under the in situ high hydrostatic pressures. However, such chemolithoautotrophs had never been cultured under the physical (particularly in pressure) and the chemical (excess solubility of gas species by pressure) conditions of their habitats until now. Therefore, their growth characteristics and physiology in the deep-sea and subseafloor habitats have been unknown.
Meanwhile, in geochemical field, origin of naturally existing methane had been estimated by means of stable carbon isotope signature (δ13) (*2) of methane (Fig.2). Based on the early experimental results of the stable carbon isotopic fractionation during the methanogenesis, the classification of origins of methane had been established. But no experiment has been done under high hydrostatic pressure condition and there has been no experimental evidence that microbes produce isotopically light methane under such conditions.
Methods
Methanopyrus kandleri strain 116(Fig.3) isolated from the Kairei hydrothermal field in the Central Indian Ridge was used in this study. The growth, survival and methane production characteristics were investigated under the in situ high-temperatures and pressures using a new technique(Fig.4 .5)).
Results
Methanopyrus kandleri strain 116 was found to grow at between 85 and 116°C under conventional culture conditions, while it became able to grow at up to 122°C under high hydrostatic pressure conditions (Fig.6). As for viability at high temperatures, it survived up to 2 hours at 130°C under normal pressure, but it could survive over 3 hours of 130°C heating under the high-pressure.
This strain synthesize cellular organic carbons by fixing CO2 while obtaining energy when producing methane from hydrogen and CO2. The stable carbon isotopic fractionations occur in both methanogenesis and carbon fixation pathways. Under conventional culture conditions, fractionation effects of methane production and carbon fixation are -27~-34 and -20~-25, respectively. They are similar to previously reported values (Fig.7). On the other hand, under high hydrostatic pressure conditions, the stable carbon isotopic fractionation becomes -10~-12, which are one of the smallest effect ever reported (Fig.8). This result shows that in the natural environment, isotopically heavy methane with about -10 of δ13C value can be produced by microorganism (Fig.8). According to the concept in geochemistry textbooks, the methane produced by strain 116 should be classified as a magmatic source (Fig.2), now that is disproved by this result.
Discussion and future prospective
Updating the upper temperature limit for life by a hyperthermophilic methanogen with a new technique suggests the chemolithoautotroph other than methanogen also elevate the limit under the same method. Surely, not only temperature but also other conditions for growth could expand the limits of life and biosphere in space as well as in Earth.
Cultivation of previously unknown microorganisms in the deep-sea and deep Earths crust will become possible by this technique. It helps largely to elucidate ecophysiological roles in the global biogeochemical processes.
Furthermore, this study has revolutionized the concept that the microbiologically produced methane is isotpically light. This study finally suggests the potential key factor to control the stable carbon isotopic fractionation during the methanogenesis. The free energy change of the methane production reaction might have a great impact on the stable carbon isotopic fractionation. This means that isotopically variable methane could be produced by microorganisms under different environmental conditions. Stable isotope geochemistry is now used broadly to investigate the co-evolution of Earth and Life through the history and the global material circulations. For the future, the refined interpretations are required based on the results though more elaborate experiments.
*1:chemoautotrophic microorganism
Microorganism grows by using inorganic redox reactions as the energy conversion sources and CO2 as the carbon source. Many gaseous energy and carbon sources (H2, H2S, CH4, CO2, CO, etc.,) are present in nature. These gaseous substances are present at extremely high concentrations in deep-sea or Earths crust environments due to the elevated solubility by pressure, so that the condition needs to be reproduced.
*2: stable carbon isotope signature (δ13C)
Carbon atom has 12,13,14 mass numbers and 13C is stable isotope and 14C is radio isotope. Substances contain carbon in nature have 12C and 13C in a certain proportion, which called stable carbon isotope ratio. Stable isotope ratio of carbon is indicated by δ13C which expressing shift range from standard specimen (belemnite in the Peedee Formation of Cretaceous in southeast USA) in permillage. In microbial metabolic system, lighter carbon atom (12C) is preferentially used so that the product tend to be light carbon-rich (δ13C shift to negative value). It is called carbon isotope fractionation by biological processes. And the degree of fractionation is called fractionation effect. The most famous example is carbon fixation by plant during the photosynthesis. Organic carbon of plant shift -20 (δ13C = approximately -30 ) compared to CO2 in the air (δ13C = approximately -10 ). In this case the fractionation effect is about -20 . It is generally believed that when microorganism produce methane from CO2, light carbon is enriched much more in generated methane than CO2 (large isotope fractionation effect). In other material circulation of elements, related with biological processes life such as hydrogen, oxygen, nitrogen and sulfur, there are many physical, chemical and biological fractionation steps. Isotope fractionation analysis is used to clarify those mechanisms.
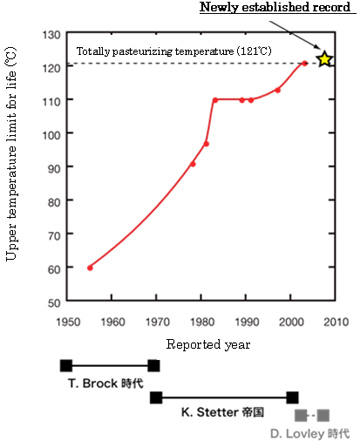
Fig.1. History of extending UTL
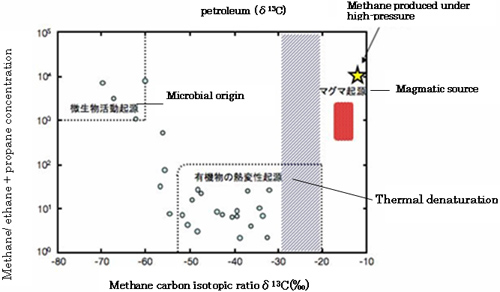
Fig.2. Classification of origins of methane in natural environments
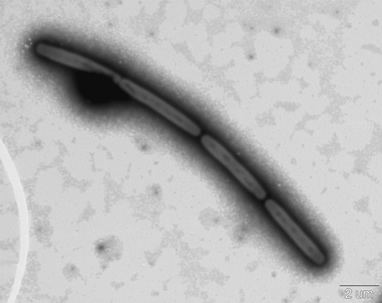
Fig.3. An electron micrograph of hyperthermophilic methanogen Methanopyrus kandleri strain 116 isolated from the Kairei Field in the Central Indian Ridge. The lower right line indicates 2 micro meters.
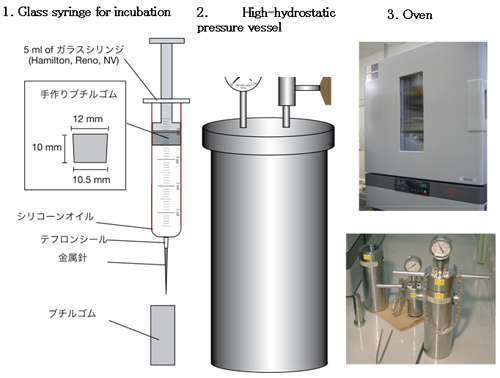
Fig.4. Tools for a newly developed high-pressure cultivation method (Takai Method) Hydraulic pressure is conveyed to gas and liquid inside of syringe by hand-made butyl rubber piston movement. Fill the empty high-hydrostatic pressure vessel with distilled water, submerge a cultivate glass syringe and apply desired pressure. And control temperature for cultivation with the oven or the incubator.
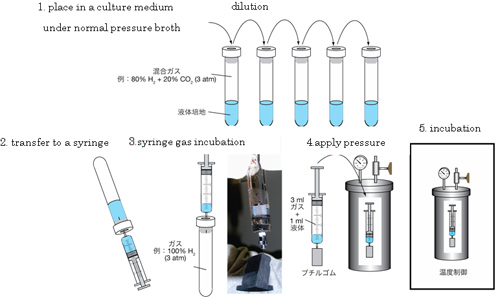
Fig.5.Procedure of Takai Method
Preparation for incubation is to be conducted using normal syringe procedure under normal pressure. Usually, microbial incubation doesnt need dilution. Dilution and incubation are conducted to obtain quantitative data from deep-sea or Earths crust samples. Inexpensive and easy-to-use Takai Method enables experiments using many more samples.
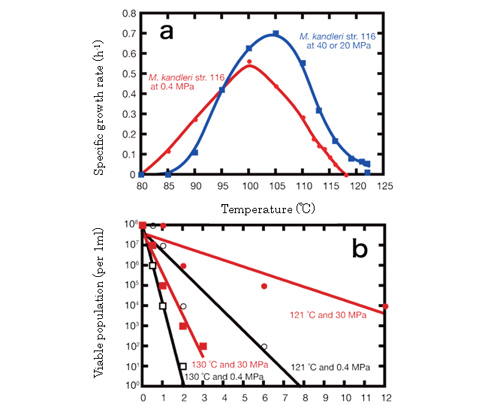
Fig.6.Temperature effects on growth and survival of hyperthermophilic methanogen Methanopyrus kandleri strain 116
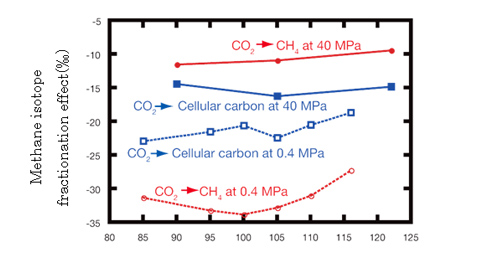
Fig.7. Stable carbon isotope fractionation under different temperatures and pressures
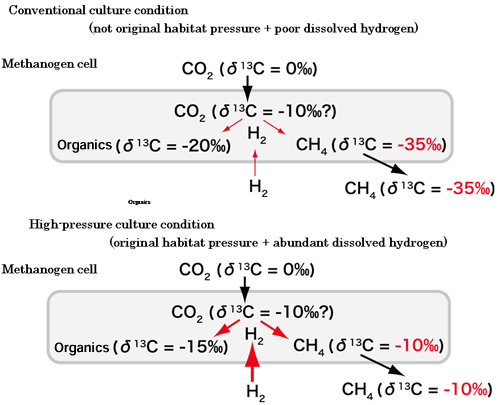
Fig.8.Carbon isotope fractionation during the methanogenesis under high-pressure culture condition
Refernces
Blöchl E, Rachel R, Burggraf S, Hafenbradl D, Jannasch HW, Stetter KO (1997) Pyrolobus fumarii, gen. and sp. nov., represents a novel group of archaea, extending the upper temperature limit for life to 113 °C. Extremophiles 1:14-21.
Kashefi K, Lovley DR (2003) Extending the upper temperature limit for life. Science 301:934.
Contacts:
The Japan Agency for Marine-Earth Science and Technology- (For the study)
Ken Takai -
Program Director, Subground Animalcule Retrieval (SUGAR) Program,
Extremobiosphere Research Center - Kenichi Tahahashi
- Manager, Research Promotion Office
- (For publications)
Noriyuki Murata, email: press@jamstec.go.jp - Manager, Planning Office Press Office